Cross-Boarder Healthcare in Life Science Tools Market South America
South America is quietly evolving into a hub for cross-border healthcare services and life science tools market. As demand for high-precision diagnostics, genomics, and biotech solutions rises, countries are increasingly outsourcing advanced lab work and clinical services across borders. In this article, we explore how cross-border healthcare is reshaping the life science tools ecosystem in South America, highlight key drivers, obstacles, and opportunities — and profile how global players like Illumina, Thermo Fisher, Qiagen and others are engaging in this dynamic landscape.
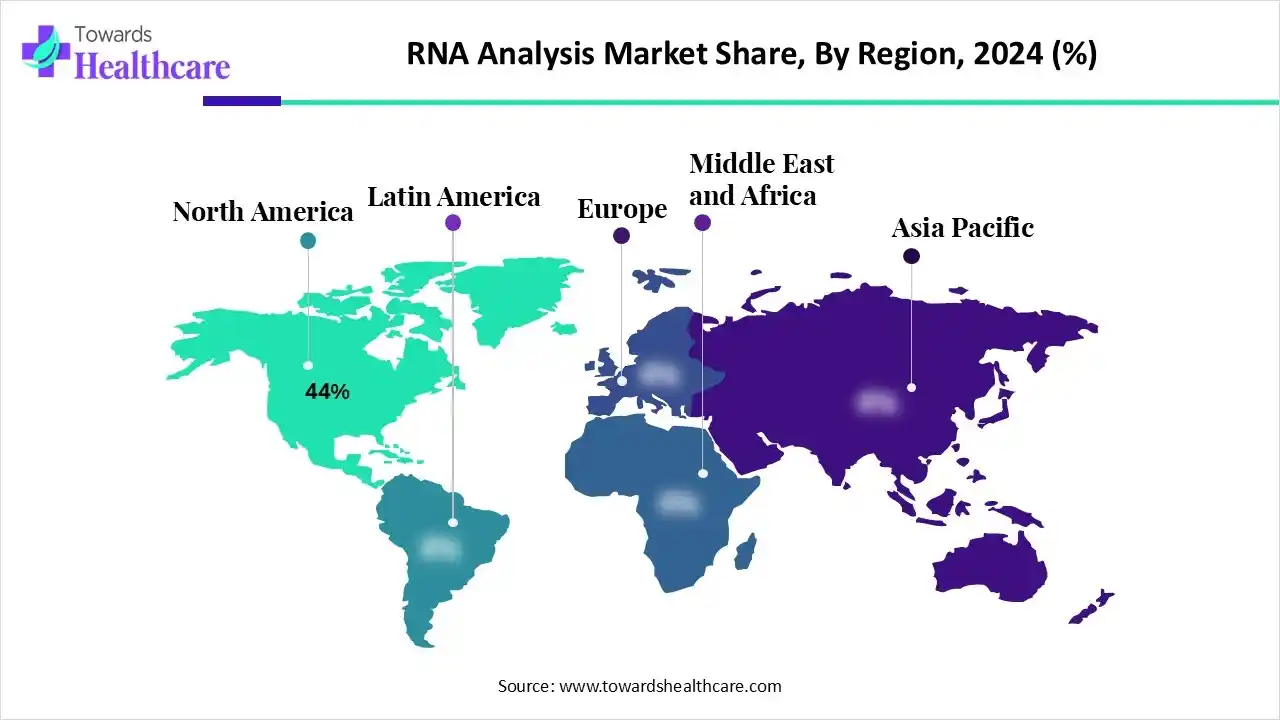
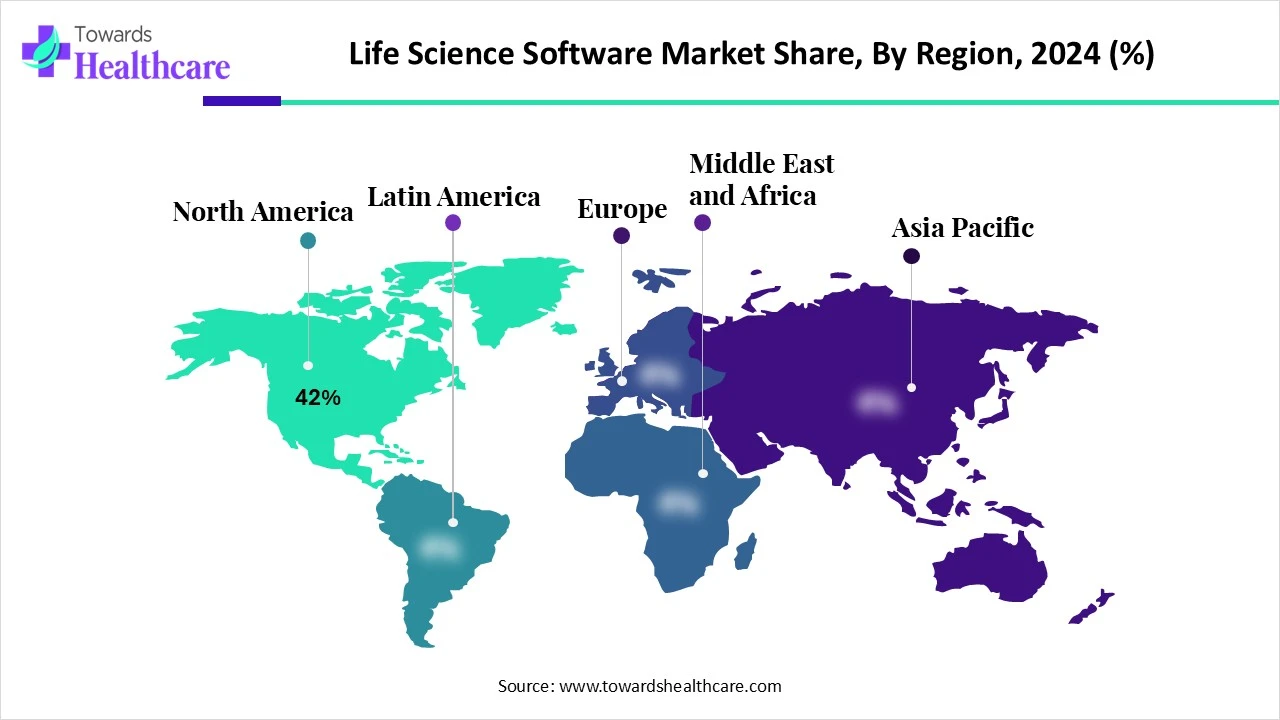
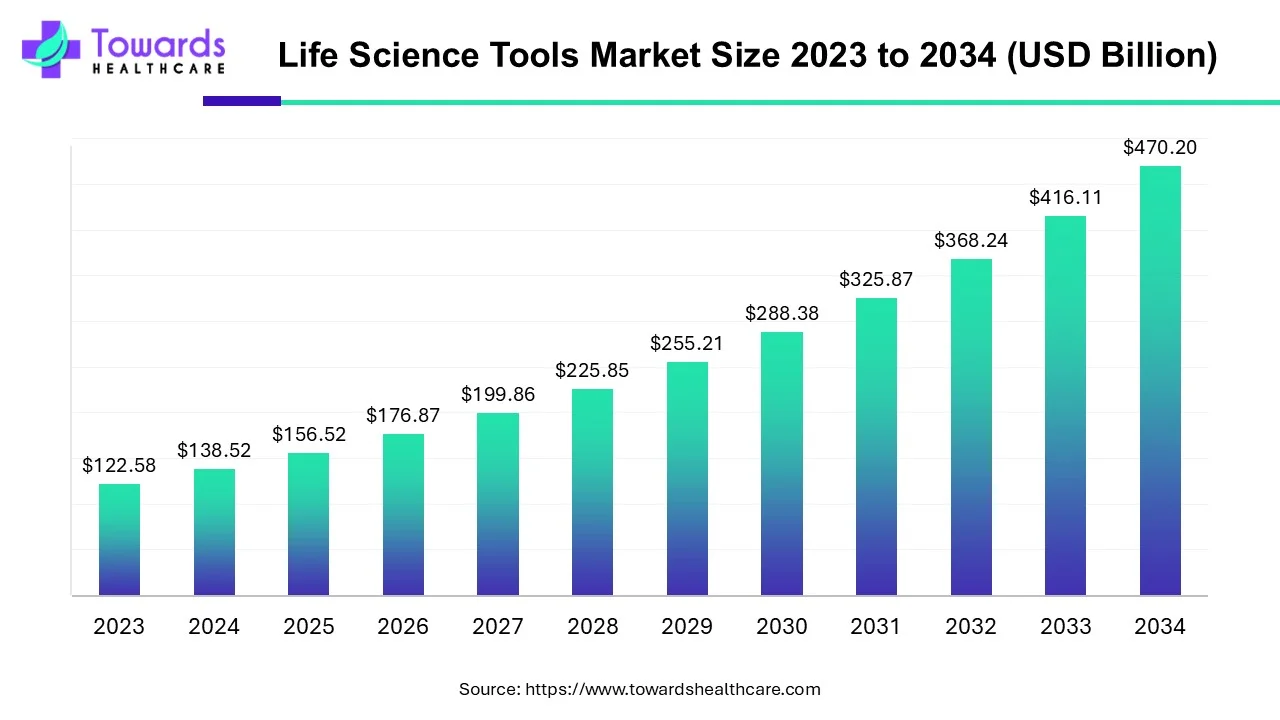
Life Science Tools Market Size
The global life science tools market size is forecast to grow at a CAGR of 13%, from USD 156.52 billion in 2025 to USD 470.20 billion by 2034, over the forecast period from 2025 to 2034, as a result of the growing R&D and technology advancements.
Download this Free Sample Now and Get the Complete Report and Insights of this Market Easily @ https://www.towardshealthcare.com/download-sample/5089
Key global players active in the region
Some life science tools and diagnostics firms already have a presence or partnerships in South America. These include:
● Illumina
● Thermo Fisher Scientific
● Qiagen
● Bio-Rad Laboratories
● Danaher
● Merck KGaA
● PerkinElmer
● Agilent Technologies
● Roche
● Becton, Dickinson & Co. (BD)
These companies supply instruments, consumables, reagents, and services to academic labs, biopharma, and diagnostic clinics across borders.
Cross-Border Healthcare Services:
Before diving deeper, let’s clarify: cross-border healthcare services here means patients, clinical samples, or specialized lab tests moving across national boundaries. Examples include:
● Sending clinical samples from a country with limited infrastructure to a neighboring country for advanced genetic testing or sequencing.
● Patients traveling to another country’s specialized hospital or lab for diagnostics or advanced therapies.
● Telemedicine / digital diagnostics enabling remote interpretation by experts in other countries.
● Shared regional lab facilities that serve multiple countries.
● This model intersects tightly with the life science tools market: the demand for advanced instrumentation, reagents, and high-end diagnostics is catalyzed by the cross-border flow of samples and services.
Drivers of Cross-Border Healthcare & Tool Adoption in South America
Infrastructure disparities and specialization gaps
Many countries in the region lack the full suite of high-end laboratory infrastructure (e.g. NGS, mass spectrometry, advanced pathology). For complex assays, sending samples across borders becomes a practical necessity.
This drives cross-border service models and demand for life science tools in better-equipped hubs.
Economies of scale & cost optimization
Pooling demand across smaller countries or institutions allows centers of excellence to invest in high-cost tools (sequencers, imaging systems) and amortize cost across clients.
This model becomes viable when national demand alone wouldn’t justify the investment.
Regulatory harmonization and regional collaboration
Initiatives to streamline regulatory approval, import/export of biological samples, and mutual recognition of diagnostic standards foster cross-border services.
Latin America has been pushing biotech collaboration, e.g., through platforms like BioLatam Digital to facilitate R&D partnerships.
Rise of precision medicine and genomics demand
The growing interest in personalized medicine, genomic profiling, and molecular diagnostics in South America forces labs to access cutting-edge tools. Not all countries can build full capacity immediately—hence cross-border lab partnerships.
This aligns well with global trends where genomics technologies are among the fastest growth segments in life science tools.
Digital transformation and remote diagnostics
With advances in telemedicine, cloud bioinformatics, AI analysis, and digital pathology, remote interpretation of data enables cross-border workflows without physical patient travel. Tools in cloud pipelines, AI diagnostics, and remote QC become crucial.
Challenges & Barriers
Regulatory, legal & sample transport constraints
Export/import of human biological samples are often heavily regulated due to biosafety, privacy, and sovereignty concerns. Customs delays, freezing logistics, and national laws (e.g. requiring in-country testing) can stifle cross-border flows.
Trust, validation & quality assurance
Receiving labs must maintain accreditation (e.g., ISO), validation, regulatory approval so that results from external labs are trusted by clinicians and payers in the originating country.
Cost, reimbursement & pricing disparity
Cross-border services incur extra logistics, tariffs, currency risk, and sometimes double handling costs. Healthcare payers may resist covering cross-border diagnostics as “out-of-network.”
Pricing models must justify the premium for advanced tools and services.
Infrastructure gaps (connectivity, cold chain, data pipelines)
Transporting samples safely, maintaining cold-chains, ensuring connectivity for data transfer and remote QC can be challenging, especially in rural or remote areas.
Competition from in-country capacity building
Governments often prefer investments in local capacity rather than outsourcing to other countries. As domestic capabilities mature, demand for cross-border services might shrink.
Strategic Opportunities & Use Cases
Regional diagnostic hubs
In countries with stable regulatory regimes (e.g. Chile, Brazil, Colombia), setting up regional centers that receive samples from less-equipped neighbors can scale well. These hubs can deploy high-end Illumina sequencers, mass spec, digital pathology and serve multiple markets.
For instance, Chile is increasingly being promoted as a Latin American health tech hub that connects local innovations to international markets.
Public-private partnerships & consortia
By combining government funding, international donors (e.g. IDB, WHO), and private life science firms, cross-border networks of labs can be built to share tools and services. The IDB Lab has historically supported health innovation projects across Latin America to widen service access.
Tele-diagnostics & digital pathology networks
Rather than physically shipping samples, slide images or genomic data can be shared across borders for expert review by pathologists or AI systems. This reduces transport risk and speeds up turnaround.
Contract research, outsourcing & shared services
Global life science tools companies (e.g. Thermo Fisher, Qiagen, Illumina) can offer cross-border sample processing or “lab-as-a-service” models, enabling smaller institutions to access high-end tools without owning them.
Focus on neglected or rare diseases
Cross-border models can help centralize rare disease diagnostics (e.g. genetic disorders) in one specialized lab for the region. Because such tests are low in volume, centralization is efficient.
How Major Players Can Leverage This Trend

1. Illumina, Thermo Fisher, Qiagen – The Genomics & Data Backbone
a. Instruments & Reagents for Regional Hubs
Illumina brings world-class next-generation sequencing (NGS) platforms. Regional hubs in Chile, Brazil, or Argentina can house Illumina’s NovaSeq or NextSeq systems, serving smaller nations that lack local infrastructure.
Thermo Fisher Scientific provides sequencing consumables, reagents, and data analysis software, ensuring continuity of services across borders. Their “end-to-end workflow” approach helps labs handle everything from DNA extraction to bioinformatics.
Qiagen is a leader in sample prep and molecular diagnostic assays, vital for ensuring consistent quality across multi-country workflows. Their QIAseq and QIAamp platforms can standardize sample handling before sequencing or PCR testing.
b. Data Portability & QC Pipelines
Cloud-enabled solutions like Illumina’s BaseSpace Sequence Hub or Thermo Fisher’s Ion Reporter software allow raw genomic data from one country to be processed, analyzed, and shared securely across borders.
QC pipelines ensure regulatory compliance — e.g., sequencing runs in Argentina can be remotely audited and validated by partner labs in Chile.
Opportunity: Together, these companies can create “Regional Genomics Alliances” in South America, offering bundled platforms (sequencers + reagents + software) and providing unified support and accreditation across borders.
2. Danaher, Bio-Rad, Agilent – The Modular & Flexible Platform Providers
a. Modular Systems for Multi-Client Labs
Danaher (through subsidiaries like Beckman Coulter, Leica Biosystems, Cytiva) offers scalable automation, flow cytometry, and proteomics platforms. Their modular nature allows one lab to serve multiple clients without dedicating machines exclusively.
Bio-Rad Laboratories adds value with digital PCR, qPCR, and protein analysis tools. These modular systems can be quickly adapted for different projects — crucial in a shared, cross-border lab model.
Agilent Technologies specializes in chromatography, mass spectrometry, and bioinformatics. Agilent’s platforms are often modular, enabling labs to expand capacity incrementally as cross-border demand grows.
b. Supporting Multi-Client Lab Networks
Shared labs in regional hubs can configure equipment depending on sample flow — e.g., shifting between oncology samples from Argentina one week and infectious disease panels from Bolivia the next.
Automation platforms (robotic liquid handlers, digital droplet PCR) reduce human error when handling high-volume, cross-border samples.
Opportunity: These companies can position themselves as “infrastructure architects”, building multi-client, multi-country labs that are flexible, scalable, and efficient.
3. Merck KGaA, PerkinElmer, Roche – The Service & Digital Integration Champions
a. Beyond Equipment: Embedding Services
Merck KGaA (MilliporeSigma) can offer integrated lab services: reagents, consumables, and digital workflow integration (e.g., electronic lab notebooks, data pipelines). Their strength lies in chemistry + biology reagents bundled with digital monitoring.
PerkinElmer provides imaging, diagnostics, and lab automation. Their “as-a-service” models allow smaller labs to lease equipment with ongoing support — perfect for cash-constrained South American markets.
Roche Diagnostics specializes in molecular and clinical diagnostics. Roche’s experience in establishing diagnostic service networks (e.g., for oncology and infectious diseases) can be replicated for cross-border models, ensuring results are trusted regionally.
b. Lab-in-a-Box & Cloud Analytics
A “Lab-in-a-Box” solution means portable, pre-configured labs (sequencing, PCR, immunoassay) that can be deployed in regional hubs or even border zones, serving multiple countries.
With cloud analytics (AI/ML), these labs can upload patient data for remote analysis by experts in Switzerland, Germany, or the U.S., while the sample stays in South America.
Roche’s NAVIFY software and PerkinElmer Signals platform already offer these cloud-based capabilities.
Opportunity: These companies can evolve into end-to-end service partners in South America — not just selling tools but running cross-border lab networks, embedding digital diagnostics, and providing subscription models.
Joint ventures or joint labs across borders: for example, a Brazilian firm partnering with an Argentine institution to share tools and diagnostics capacity.
Narrative Case Example (Illustrative)
Suppose a hospital in Bolivia lacks a next-generation sequencing (NGS) lab. It sends patient samples across the border to a genomics hub in Chile. The Chilean lab uses Illumina sequencers to run whole-genome analysis and sends back results via a secure digital portal. The Bolivian clinicians use that data for precision oncology decisions. Over time, the network scales to include Paraguay, Peru, and northern Argentina, with cost-sharing models and regional reimbursement agreements.
Within this network, Chile becomes a service hub, attracting instrument suppliers like Thermo Fisher, Qiagen, and Agilent to locate local distribution or even mini production. Government backing and regulatory harmonization at the Andean community level facilitate sample flows and accreditation recognition.
Recommendations & Strategic Imperatives
-
Harmonize regulatory and ethical frameworks
Encourage cross-border agreements recognizing lab accreditation, streamline sample import/export rules, and harmonize privacy laws to allow seamless data flow. -
Invest in regional centers of excellence
Governments and consortia should co-invest in diagnostic hubs (NGS, advanced imaging) that serve multi-country demand rather than duplicating infrastructure in each country. -
Adopt digital and remote pipelines
Leverage telepathology, cloud genomics, AI-based diagnostics to reduce the need for physical sample transfer. -
Innovate pricing/sharing models
Create tiered or subscription-based models, cost-sharing among clients, and provider-of-service agreements to make cross-border services financially viable. -
Engage global life science tools firms as partners, not just vendors
Encourage those firms to invest in local training, support, service hubs, supply chain presence, and shared lab models. -
Raise visibility & trust via pilot projects
Run flagship cross-border projects (e.g. rare disease panels, cancer genomics) to build confidence among clinicians, payers, and regulators.
Top 5 FAQs
1. What is driving growth in the South America life science tools market?
Growth is fueled by rising cross-border healthcare demand, increasing genomic research, regional lab hubs, and the adoption of cloud-enabled diagnostic platforms.
2. How do global companies like Illumina, Thermo Fisher, and Roche impact the market?
These companies supply advanced sequencing systems, modular lab platforms, and digital service models that enable multi-country healthcare networks.
3. Why are cross-border healthcare services important in South America?
They allow smaller nations to access advanced diagnostics and genomics through regional hubs, ensuring quality care without building costly local infrastructure.
4. What opportunities exist for life science tool providers in the region?
Opportunities include establishing shared labs, offering lab-as-a-service models, embedding cloud analytics, and creating scalable “lab-in-a-box” solutions.
5. What challenges could slow adoption of life science tools in South America?
Key challenges include regulatory differences, uneven infrastructure, high equipment costs, limited skilled workforce, and data-sharing concerns across borders.
Access our exclusive, data-rich dashboard dedicated to the life science sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/price/5089
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest