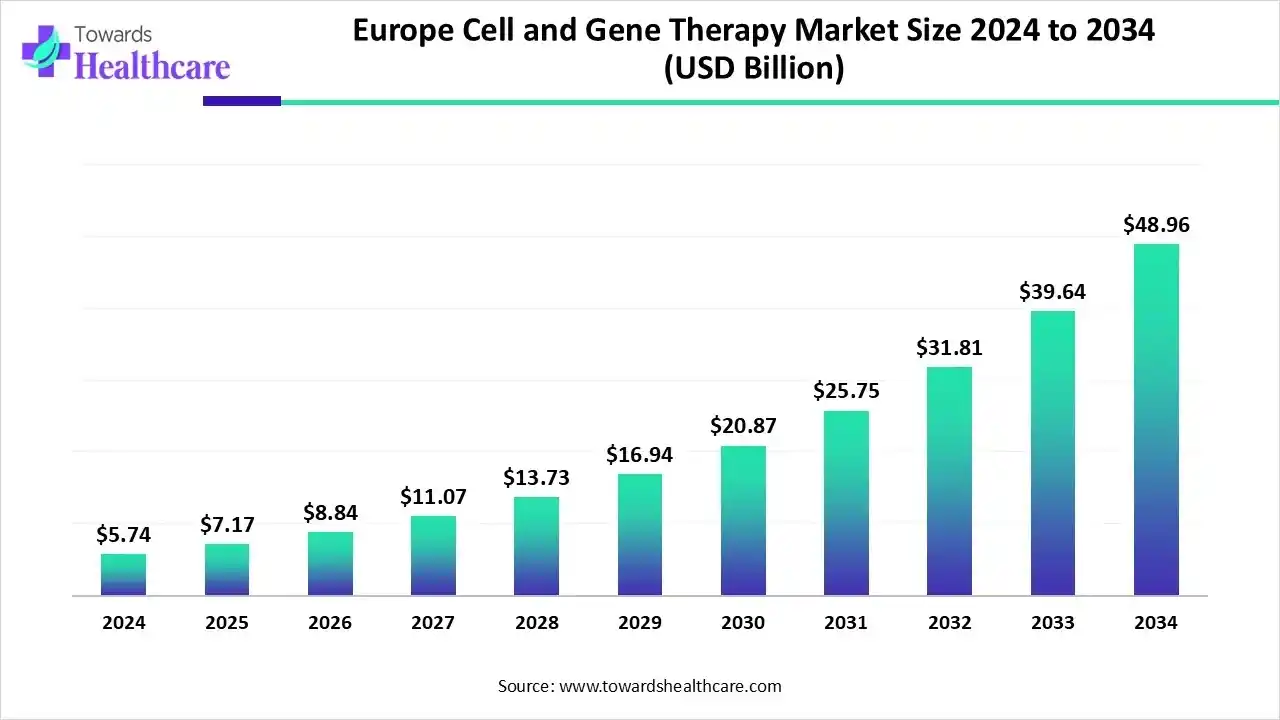
Europe cell and gene therapy market is US$2.74B in 2024, set to reach US$7.17B in 2025 and US$48.96B by 2034 at a 23.90% CAGR (2025–2034), propelled by EMA-backed approvals, Horizon Europe funding, and rapid uptake of CAR-T.
Download this Free Sample Now and Get the Complete Report and Insights of this Market Easily @ https://www.towardshealthcare.com/download-sample/6295
Market Size (in-depth, point-wise)
●2024 base: US$2.74B; demand anchored by cell therapy (58% share), viral vectors (72%), autologous cell source (62%), clinical-scale production (67%), and biopharma/biotech end users (52%).
●Near-term step-up (2025): US$7.17B with pipeline conversions and initial scale-up of allogeneic platforms; gene therapy enters fastest-growth phase (2025–2034).
●Long-term projection (2034): US$48.96B, reflecting shift from clinical-scale → commercial-scale as closed/automated systems mature.
●Compound growth: 23.90% CAGR (2025–2034) supported by oncology dominance (~48% 2024) and rare disease acceleration.
●Regional anchor: Germany 28% share (2024); leadership reinforced by a national GCT strategy and expanding vector capacity.
●Capital flows (2025 highlights): SpliceBio US$135M Series B; HAYA Therapeutics US$65M Series A; Laverock >£20M seed+—fueling platform and pipeline build-out.
●Policy tailwinds: EMA/CHMP multi-asset positive opinions (Aug-2025); EU Green Deal and proposed EU Biotech Act lower scale-up frictions and favor greener bioprocessing.
●Modality mix shift: Autologous → Allogeneic growth for throughput and cost; non-viral vectors accelerate on safety/cost manufacturing advantages.
●Use-case concentration: Oncology remains the adoption beachhead; rare genetic disorders lead future growth via curative intent.
●Manufacturing inflection: Transition from manual, variable clinical workflows to automated, standardized commercial platforms unlocks capacity and reduces COGS.
Market Trends
●Regulatory momentum: EMA/CHMP recommended 10 new medicines + 8 biosimilars (Aug-2025); multiple label expansions—shortening time to market.
●Funding scaffolding: Horizon Europe and national programs de-risk trials, especially for CAR-T and rare disease assets.
●Green manufacturing push: EU Green Deal drives closed-loop, energy-efficient bioprocessing; vendors differentiate on sustainability metrics.
●Hospital-based manufacturing pilots: EASYGEN consortium (Aug-2025; €8M; Fresenius-led) targets few-day personalized cell therapy turnaround.
●Strategic M&A/CDMO consolidation: Formation of Minaris Advanced Therapies and vector-scale deals (e.g., SK pharmteco LVV 200L CGMP batch in Paris ecosystem).
●Vector tech bifurcation: Viral vectors (72% 2024) dominate; non-viral (LNPs, physical methods) surge on immunotoxicity and cost advantages.
●Autologous today, allogeneic tomorrow: Autologous 62% (2024) share; allogeneic pipelines scale with universal donor strategies.
●Oncology first, rare disease next: Oncology ~48% (2024); rare genetic disorders expected fastest growth on single-gene correction potential.
●AI permeation: From design → QC → release (details below) improving yields, safety, and time-to-patient.
●Commercial-scale readiness: Clinical-scale 67% (2024) now; the commercial-scale segment becomes the fastest-growing through 2034.
10 Deep Ways AI Impacts This Market
●In-silico vector design: AI optimizes AAV capsids/LVs for tropism, payload, and immunogenicity trade-offs, raising effective dose success odds.
●Promoter/construct optimization: ML models score promoters, enhancers, and UTRs for expression stability across tissues and patients.
●Manufacturing set-point control: Reinforcement learning tunes MOI, cell density, and feed strategies to maximize vector yield and CQAs.
●Automated eBR & deviation analytics: NLP on batch records detects anomaly patterns, cutting batch failure rates and CAPA cycles.
●Rapid QC release: Computer vision + ML for sterility, identity, potency proxies to accelerate lot release without compromising safety.
●Patient stratification: Multi-omics fusion models select responders for CGT, reducing trial size and improving probability of technical success.
●Safety prediction: Off-target/oncogenicity risk scoring for gene editing; immunogenicity forecasts to guide premedication/regimens.
●Digital twins for scale-up: Virtual bioreactor twins translate process from 2L→200L with fewer experiments and higher transfer fidelity.
●Hospital workflow orchestration: Scheduling/throughput AI shortens vein-to-vein times for CAR-T; predicts bed/OR and apheresis slots.
●Post-market pharmacovigilance: Signal detection on EHR/registry data identifies long-term AEs, informing label updates and REMS.
Regional Insights
Germany (28% 2024; leader)
●Policy & strategy: National GCT strategy via BMBF/BIH coordination; clear hospital-academia-industry interfaces.
●Manufacturing muscle: LVV/AAV capacity builds (e.g., 200L CGMP LVV initiatives), strong automation vendors.
●Clinical density: High trial throughput in oncology and rare disease; reimbursement pilots for ATMPs.
United Kingdom
●Company cluster: Oxford Biomedica, Orchard, Autolus, Adaptimmune, GSK, AstraZeneca drive end-to-end ecosystem.
●Translational hubs: Catapult centers expedite process development and scale-up.
●Policy continuity: MHRA/EMA alignment pathways for EU access via partnerships.
France
●Innovation nodes: Sanofi, Cellectis, GenSight; strong gene-editing pedigree.
●Public funding: Robust academic networks; expanding hospital-based production pilots.
●Therapy focus: Oncology, ophthalmology, and metabolic indications.
Italy
●Legacy strengths: MolMed heritage and hospital-linked manufacturing; expanding rare disease programs.
●CDMO linkages: Access to pan-EU vector networks; growing GMP talent pool.
●Regulatory support: Streamlined ATMP approvals via national competence centers.
Spain
●Trial growth: Rising AAV ophthalmology and hematology trials.
●Cost advantage: Competitive bioprocessing OPEX; strong academic consortia.
●Infrastructure: Upgrades to closed systems improve batch consistency.
Nordics (Sweden/Denmark/Norway)
●Precision medicine focus: Registry depth enables long-term outcomes capture.
●Bioprocess innovation: Sustainable, low-footprint facilities aligned with Green Deal.
●Access pathways: Early patient access schemes for rare diseases.
Netherlands
●uniQure anchor: AAV heritage and commercial know-how.
●Logistics: Central EU location for expedited distribution.
●Tech transfer: Strong university-industry patenting pipeline.
Market Dynamics
Drivers
●EMA/CHMP approvals and supportive CAT pathways; Horizon Europe and national grants.
●Oncology adoption (48% 2024) and CAR-T popularity; Germany 28% regional engine.
●Capital inflows (SpliceBio US$135M, HAYA US$65M, Laverock >£20M).
Restraints
●High COGS and variable yields (autologous); limited vector capacity bottlenecks.
●Complex QA/QC release and vein-to-vein logistics.
●Reimbursement uncertainties for ultra-rare indications.
Opportunities
●Allogeneic and non-viral platforms for cost/scale breakthroughs.
●Green bioprocessing differentiation; hospital-based rapid manufacturing (EASYGEN).
●AI-driven process control and safety—shorter cycles, higher success rates.
Challenges
●Talent shortages in GMP/automation; cross-border regulatory variability.
●Long-term safety surveillance requirements.
●Transition risk from clinical (67%) → commercial scale without disrupting supply.
Top 10 Companies
Oxford Biomedica (UK)
●Products: LVV development & manufacturing; platformed CAR-T/CAR-NK vectors.
●Overview: Premier EU lentiviral CDMO with end-to-end capabilities.
●Strength: Deep LVV IP, quality track record, large-scale suites.
Orchard Therapeutics (UK)
●Products: Ex vivo autologous gene therapies for rare inherited disorders.
●Overview: Focused on hematopoietic stem cell gene therapy.
●Strength: Curative intent data; pediatric rare disease expertise.
Autolus Therapeutics (UK)
●Products: Next-gen CAR-T programs for hematologic malignancies.
●Overview: Modular CAR designs with safety switches.
●Strength: Engineering sophistication; manufacturing control.
GlaxoSmithKline – GSK (UK)
●Products: Oncology/rare disease CGT portfolio; precision therapeutics expansion.
●Overview: Big pharma scale, BD-driven pipeline (e.g., 2025 precision dealmaking).
●Strength: Global development, market access, and PV infrastructure.
AstraZeneca (UK/Sweden)
●Products: In vivo cell therapy platforms; ENaBL lentiviral nanobody approach.
●Overview: Oncology-centric CGT with novel delivery.
●Strength: Broad oncology footprint; rapid clinical translation.
Sanofi (France)
●Products: Gene therapies and immunology focused programs.
●Overview: Integrated research, manufacturing, and market access.
●Strength: Late-stage development engine and global reach.
Cellectis (France)
●Products: Allogeneic gene-edited cell therapies (TALENs).
●Overview: Pioneering gene-editing platform company.
●Strength: Off-the-shelf ambition; editing know-how.
Miltenyi Biotec (Germany)
●Products: Cell processing systems (CliniMACS), reagents, and tools.
●Overview: Enabling technology leader across CGT workflows.
●Strength: Ubiquitous footprint in GMP cell processing.
Evotec (Germany)
●Products: End-to-end discovery to CMC services; RNA/cell therapy platforms.
●Overview: Partner-centric model with EU/US facilities.
●Strength: Scale, data platforms, and BD network.
uniQure (Netherlands)
●Products: AAV gene therapies targeting monogenic diseases.
●Overview: AAV pioneer with clinical and commercial experience.
●Strength: Vector platform depth; manufacturing expertise.
Latest Announcements
AstraZeneca (Mar-2025): Definitive agreement to acquire EsoBiotec to advance in vivo cell therapies using ENaBL—aims for minutes-scale delivery vs current complex workflows.
BioIVT (Oct-2025): Showcased expanded cellular solution portfolio across major CGT conferences—broader access to high-quality inputs for R&D/QC.
ABALETTA Bio (Jun-2025): Released new clinical/translational data for rese-cel in autoimmune indications (RESET programs), broadening CGT applicability.
Minaris Advanced Therapies (May-2025): Formed by combining Minaris Regenerative Medicine with WuXi AT (US/UK ops) under Altaris—global CDMO scale-up.
SK pharmteco Cell & Gene Europe / AP-HP / Imagine (Feb-2025): Contract for 200L CGMP LVV clinical batch and regulatory support—EU LVV capacity uplift.
EMA/CHMP (Aug-2025): Positive opinions for 10 new medicines, 8 biosimilars, plus 4 generics and multiple new indications—pipeline acceleration.
EASYGEN (Aug-2025): €8M EU-backed hospital-based automated platform for personalized cell therapies (Fresenius-led).
European Commission (Jul-2025): Long-term life sciences strategy; proposed EU Biotech Act—policy momentum for CGT scale-up.
Recent Developments
●Consolidation and capacity build: Minaris Advanced Therapies creation; SK pharmteco LVV 200L run in Paris ecosystem.
●Capital rounds energizing pipelines: SpliceBio (US$135M), HAYA (US$65M), Laverock (>£20M).
●Oncology lead use-case: CHMP opinions + CAR-T popularity sustain oncology dominance (~48% 2024).
●Sustainability shift: EU Green Deal catalyzes closed-loop, low-energy CGT manufacturing adoption.
●AI integration: Workflow automation from construct design → PV improving cycle times and QA.
Segments Covered
Therapy Type
Cell Therapy (58% share, 2024)
Modalities & use-cases
●Immune cell therapies: CAR-T (hematologic cancers), TCR-T (solid/viral antigens), NK cells (off-the-shelf potential).
●Stem/progenitor cells: MSCs for immunomodulation; HSCs for ex-vivo gene correction; iPSCs for scalable allogeneic banks.
Why it leads today
●Clinically validated paths: Multiple EMA-approved CAR-T lines; clearer CMC and hospital workflows.
●Predictable outcomes in oncology: Rapid measurable responses; defined endpoints (ORR, MRD negativity, PFS).
Manufacturing & logistics
●Autologous dominance (see below): Apheresis → engineering → release; vein-to-vein orchestration is critical.
●Key bottlenecks: Viral vector supply, QC release time, fresh vs cryo chain reliability.
Cost & quality levers
●Closed systems, automated cell selection/activation, real-time analytics to cut batch failures and COGS.
Near-term evolution
●Scaleout to more centers; allogeneic NK/iPSC-derived cells to reduce lead times and cost per dose.
Gene Therapy (fastest growth 2025–2034)
Modalities
●Gene augmentation: Functional copy delivery for monogenic disease.
●Gene editing: CRISPR/TALENs/ZFNs for precise correction or knockout.
●RNA-based: mRNA for transient protein expression; siRNA/ASO for gene silencing.
Why it accelerates
●Curative intent in rare disorders; maturing vector platforms; expanding payer experience with outcomes-linked models.
CMC focus
●Vector productivity (capsid design, promoter strength), purity (empty/full capsid ratios), robust analytics (potency, biodistribution).
Clinical design trends
●Basket trials by genotype; long-term follow-up plans for durability and late AEs.
Therapeutic Area
Oncology (48% in 2024)
Scope
●Hematologic (ALL, DLBCL, MM), pediatrics, and increasingly solid tumors (gating antigen selection, TME modulation).
Why it dominates
●Clear surrogate endpoints; hospital infrastructure for apheresis/infusion; strong clinician familiarity.
What’s next
●Multiplex edits (armor, knock-outs), logic-gated CARs, combination with checkpoint inhibitors.
Rare Genetic Disorders (fastest CAGR)
Scope
●Hematology (hemophilia, hemoglobinopathies), neuromuscular, ophthalmology, metabolic diseases.
Growth drivers
●Single-gene targets, registries enabling rapid identification, advocacy-led trial recruitment.
Market access
●Outcomes-based contracts, newborn screening linkages, long-term evidence capture via national registries.
Others (Neuro, Cardio, Ophthalmology, Metabolic/Endocrine, Infectious)
Neuro: AAV to CNS (intrathecal/intraparenchymal); challenge = immune responses, dosing windows.
Cardio: Regenerative cell therapy for ischemia/heart failure; endpoints require longer follow-up.
Ophthalmology: Local delivery, immune privilege, sensitive functional endpoints (BCVA, FST).
Metabolic/Endocrine: Liver-tropic AAV/LNP; lifelong durability and re-dosing strategies are key.
Infectious: Engineered immune cells/antibodies for refractory infections; hospital stewardship pathways matter.
Vector Type (Gene Therapy)
Viral Vectors (72% in 2024)
Families & fits
●AAV: Non-integrating, durable expression; favored for eye, liver, muscle.
●Lentivirus (LV): Integrating; gold standard for ex-vivo HSC/immune cell modification.
●Retrovirus/Adenovirus: Niche uses; adenovirus for high payload but immunogenic.
Why dominant
●High transduction efficiency, established QC frameworks, known toxicology.
Manufacturing priorities
●Upstream yield (HEK293/suspension), downstream purification (chromatography), accurate titering (ddPCR).
Risks to manage
●Immunogenicity (pre-existing NAbs), insertional mutagenesis (integrating vectors), dose-related toxicity.
Non-Viral Vectors (fastest growth)
Approaches
●LNPs: mRNA/siRNA delivery; scalable and re-dosable.
●Physical methods: Electroporation for ex-vivo cell engineering (CAR-T/NK).
Why they’re rising
●Lower immunotoxicity, simpler scale-up, reduced COGS, flexible payloads.
Challenges
●Tissue tropism, endosomal escape, durability—addressed through lipid chemistry and targeting ligands.
Cell Source (Cell Therapy)
Autologous (62% in 2024)
Strengths
●Minimal graft-versus-host, high response in hematologic cancers, well-trodden regulatory path.
Constraints
●Patient fitness, long lead times, batch-of-one economics, site-to-site variability.
Improvements
●Point-of-care automation, standardized release assays, cryo logistics optimization.
Allogeneic (fastest growth)
Value proposition
●Off-the-shelf inventory, immediate availability, lower per-dose cost potential, easier scale economics.
Technical keys
●Gene edits to evade host immunity (HLA, CD52), graft persistence, reduced rejection.
Use-cases
●First in NK/iPSC-derived products; expansion toward T-cell therapies and regenerative indications.
Manufacturing Scale
Clinical-Scale (67% in 2024)
Characteristics
●Small-to-mid bioreactor volumes; semi-manual steps; extended QC turnaround; site-specific SOPs.
Pain points
●Batch variability, high labor burden, tech-transfer friction between sites, lengthy vein-to-vein times.
What’s changing
●Adoption of closed systems, electronic batch records, and automated analytics to raise right-first-time.
Commercial-Scale (fastest growth)
Defining features
●Standardized, automated, high-containment suites; platform processes; global release testing networks.
Benefits
●Lower COGS, higher throughput, reproducible CQAs, improved access for larger patient cohorts.
Readiness factors
●Robust comparability packages, global supply chain resilience, sustainability metrics (energy/water).
End User
Biopharma & Biotech (52% in 2024)
Roles
●Discovery, vector engineering, IND/MAA ownership, CMC platforming, market access execution.
Needs
●Reliable vector and plasmid supply, rapid process development, partnerable CDMO capacity.
Metrics
●Probability of technical/regulatory success, time-to-first-in-human, COGS/dose, lot release cycle time.
Hospitals & Specialty Clinics (fastest growth)
Why growing
●Expansion of certified infusion/manufacturing centers; near-patient production pilots; integrated care pathways.
Operational focus
●Apheresis scheduling, cell handling SOPs, AE management, long-term follow-up.
Key enablers
●Point-of-care platforms, reimbursement frameworks, digital orchestration of vein-to-vein.
Academia & Research Institutes; CMOs/CROs
Academia/Institutes
●Early discovery, preclinical models, first-in-human trials, translational know-how.
CMOs/CROs
●Capacity bridging, tech-transfer, global QC/release, regulatory documentation support.
Strategic value
●Flex capacity during surges; specialized analytics; faster scale transitions.
Region (Europe)
Germany (≈28% share, 2024)
Why leader
●National GCT strategy; dense clinical networks; strong vector and device ecosystems.
Focus areas
●Oncology and rare diseases; expansion of GMP vector volumes (e.g., 200L LVV clinical batches).
System strengths
●Reimbursement pilots for ATMPs, high-quality registries, engineering talent base.
United Kingdom
Industrial base
●Oxford–London–Cambridge triangle with Oxford Biomedica, Orchard, Autolus, Adaptimmune, GSK, AstraZeneca.
Translational assets
●Catapult centers, strong tech-transfer culture, hospital manufacturing initiatives.
Access
●Rapid setup for first-in-human; EU access via partnerships and aligned standards.
France
Players
Sanofi, Cellectis, GenSight; strengths in editing and ophthalmology.
Infrastructure
●Public funding, Paris hospital networks, academic-clinical consortia.
Trajectory
●Allogeneic editing and ophthalmic AAV scale-up.
Italy
Legacy competency
●Longstanding hospital-linked manufacturing (e.g., ex-MolMed lineage).
Focus
●Oncology, rare disease; increasing CDMO partnerships.
Enablers
●Competence centers streamlining ATMP approvals and trials.
Spain
Momentum
●Ophthalmology and hematology AAV trials rising; cost-competitive GMP operations.
Network
●University-hospital consortia; upgrades to closed systems for consistency.
Opportunity
●Regional hubs for Southern Europe distribution.
Nordics (Sweden, Denmark, Norway)
Unique assets
●Population registries for longitudinal outcomes; strong clean-tech alignment.
Focus
●Precision medicine pilots, early access for rare disease programs.
Scale
●Boutique but high-quality manufacturing and data infrastructure.
Netherlands
Anchor
●uniQure AAV heritage; experienced in commercial pathways.
Logistics
●Central EU distribution, port access, efficient customs for cold chain.
R&D
●University-industry patenting and rapid clinical onboarding.
Top 5 FAQs (using given data)
-
What is the market size now and where is it heading?
US$2.74B (2024) → US$7.17B (2025) → US$48.96B (2034) at 23.90% CAGR (2025–2034). -
Which therapy type leads today?
Cell therapy (58% share, 2024); gene therapy is the fastest-growing through 2034. -
Which therapeutic area is largest?
Oncology (48% 2024); rare genetic disorders expected to grow fastest. -
What vectors and cell sources dominate?
Viral vectors (72% 2024) and autologous cells (62% 2024); non-viral and allogeneic are fastest-growing. -
Which region leads in Europe?
Germany (28% share, 2024) on strong R&D, policy, and manufacturing initiatives.
Access our exclusive, data-rich dashboard dedicated to the therapeutic area sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Checkout this Premium Research Report @ https://www.towardshealthcare.com/checkout/6295
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest